RAW Protein Part I: Protein Structure and Amino Acids

Protein has been established as one of the most significant subjects to include when discussing nutrition and health. Any fitness professional will suggest tracking protein type and intake, and the pet owners are starting to do the same for their animals. For those feeding a raw diet, it’s essential to understand how consuming protein influences our overall health right down to the cellular level. When it comes to choosing protein sources for our pets, the main goal is meeting their essential amino acid requirements provided by organizations such as the NRC (National Research Council).
What is Protein?
We consume various protein types in our diet through different foods, like meat, fish, and beans; it’s also one of the most abundant macromolecules in our body. Not all protein is created equally, nor does it have a single purpose. While it’s common to hear protein is essential for building muscle, it’s also vital for organ, tissue, and cell functions. Simply, “a primary function of dietary protein is as a source of essential amino acids and nitrogen for the synthesis of nonessential amino acids” (Sanderson, 2013).Some examples of protein found in our body include dietary enzymes, regulatory hormones, and cell structure components. Without protein, we wouldn’t have proper cell structure or communication, among other functions that are essential to life.

A protein’s molecular structure dictates its function and purpose. Amino acid chains, known as peptides, are crucial to cellular function. Proteins can be thousands of amino acids long where others are smaller than 100 amino acids in length. Working proteins are found in these different structural levels throughout our bodies. Here we observe protein from the most complex to basic structures: quaternary, tertiary, secondary, and primary. We breakdown protein to understand how the smallest unit influences the entire protein.
Protein Structure Basics

The quaternary structure of a protein consists of multiple subunits that are joined through molecular interactions. Human hemoglobin (Figure 1) is a classic example of a functional protein found in a quaternary final structure, as 2 alpha-helixes and 2 beta-sheets complete the 4 subunits of the protein. It should be noted that quaternary formation is not needed to form a functional protein. Yet, many biologically active proteins are found in this structure.
Next, the tertiary structure consists of multiple polypeptides (peptides larger than 100 amino acids) interacting with each other through intermolecular forces (between separate molecules). This gives a polypeptide it’s characteristic folded appearance. For example, insulin is an important hormone, secreted from the pancreas, to regulate blood sugar levels; it’s found naturally in a tertiary structure. Each interaction between polypeptides dictates the turns and twists found in a tertiary structure.

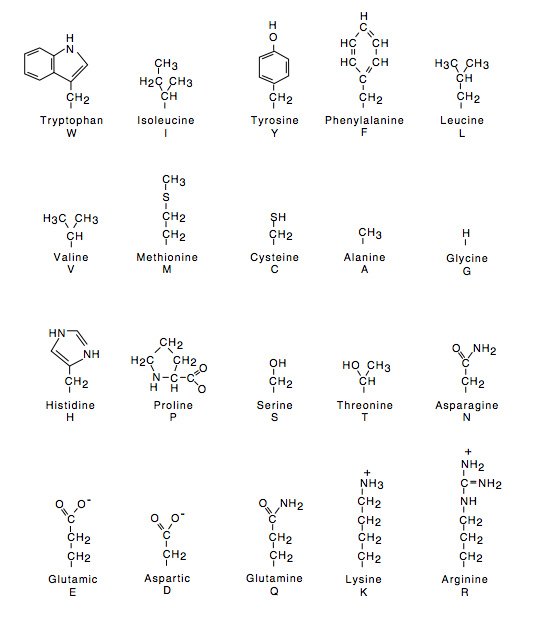
Polypeptides form two types of secondary structures: an alpha-helix, or a beta-sheet. Each unit within the peptide interacts with side groups found on other units, to create a helical twist or a flat sheet. Similar to forces acting in tertiary structures, the intramolecular forces found in a peptide are based on the unique combination of side groups for each protein. The primary structure is a peptide – connecting amino acids with a peptide bond. Amino acids feature a base with distinguishing side-chains, as shown in Figure 5—interactions between side-chains cause folding needed for functionality.
Amino Acids
Now, molecular forces are mentioned repeatedly in quaternary, tertiary and secondary protein structures. These chemical interactions are based on the unique side-chains found on different amino acids. The different combinations of amino acids that are bonded lay the foundation for the overall structure from primary to quaternary levels. Meeting amino acid requirements is essential to catabolizing functional protein molecules.
Essential vs Non-Essential Amino Acids
Not all amino acids need to come through diet. Specific genes in an organism’s genome allow for the production of some amino acids through metabolic processes. These amino acids are regarded as non-essential since a body can produce sufficient amounts through metabolism and catabolism processes alone. Amino acid catabolism is unique to each species, just as each creature has a unique digestive system.
Human Requirements
| Amino Acid | Daily Min. (g/kg) |
|---|---|
| Histidine | 0.010 |
| Leucine | 0.020 |
| Isoleucine | 0.039 |
| Lysine | 0.030 |
| Methionine + cysteine | 0.015 |
| Phenylalanine + tyrosine | 0.025 |
| Threonine | 0.015 |
| Tryptophan | 0.004 |
| Valine | 0.026 |
Canines
| Amino Acid | Daily Min. (g/1000kcal) |
|---|---|
| Arginine | 0.70 |
| Histidine | 0.37 |
| Isoleucine | 0.75 |
| Leucine | 1.35 |
| Lysine | 0.70 |
| Methionine + cysteine | 1.30 |
| Phenylalanine + tyrosine | 1.48 |
| Threonine | 0.85 |
| Tryptophan | 0.28 |
| Valine | 0.98 |
However, the amino acids they can’t produce through cell processes are known as essential amino acids. These must be ingested through food matter, and can’t be created within the body alone. Missing these vital nutrients will diminish the body’s overall function, as we noticed earlier where all protein is reliant on the correct combination of amino acids.
How do Amino Acids Influence Protein Sources?
A diet consisting of varied protein sources is vital to meeting the essential amino acids your dog needs. We need different amino acids (Table 1) than our dogs (Table 2). Formulating a raw diet requires thoughtful consideration for the variant amino acid profiles of common meats like beef, chicken, and pork. To maintain good health for our dog on a raw diet, essential (and non-essential) amino acids must be met to ensure proper protein function. It’s critical not to take human dietary information and apply it to our pets, or crucial nutrition will be missed while formulating a raw diet for your dog.

Protein is an incredibly important biomolecule and energy source. The amino acid content in the meat of common livestock species is not the same across all. For example, similarily weighed cuts from beef and pork will not contain the same amino acid profile. Meeting intake requirements is vital for overall health and bodily function, from the primary peptide to complex quaternary proteins that act as enzymes, hormones, and other regulatory biomolecules.
Part 2 will extend on our knowledge of amino acid requirements to correctly source different animal proteins to meet minimum protein requirements for your dog. Stay tuned!
References
- Alberts B., Johnson A., Lewis J., et al. (2002). Molecular Biology of the Cell, New York: Garland Science, 4th Ed.
- Dasgupta, J., Sen, U., Choudhury, D., Dutta, P., Basu, S., Chakrabarti, A., Chakrabarty, A., Dattagupta, J.K. (2003). Crystallization and preliminary X-ray structural studies of hemoglobin A2 and hemoglobin E, isolated from the blood samples of β-thalassemic patients. Biochemical and Biophysical Research Communications, 303(2):619-23. DOI: 10.1016/S0006-291X(03)00379-6
- OpenStax. (2013). Anatomy and Physiology, OpenStax CNX. Retrieved from http://cnx.org/contents/14fb4ad7-39a1-4eee-ab6e-3ef2482e3e22@8.24
- Sanderson, S. (2013). Nutritional Requirements and Related Diseases of Small Animals. Merick Manual Veterinary Manual,
- Shleif, R. (1993). Genetics and Molecular Biology, The Johns Hopkins University Press: Baltimore, Maryland, 2nd Ed. ISBN: 0-8018-4673-0
- Sreekanth, R., Pattabhi, V., Rajan, S.S. (2008). Structural interpretation of reduced insulin activity as seen in the crystal structure of human Arg-insulin. Biochemie, 90(3):467-73. DOI: 10.1016/j.biochi.2007.09.012
- The World Health Organization. (2007). Protein and Amino Acid Requirements in Human Nutrition. WHO Technical Report Series, no. 935. ISBN: 92 4 120935 6